|
3/21/2024 0 Comments Bohr's model of atom Thus, the columns of the periodic table represent the potential shared state of these elements' outer electron shells that is responsible for their similar chemical characteristics. When an atom gains an electron to become a negatively-charged ion this is indicated by a minus sign after the element symbol for example, \(F^-\). Group 17 elements, including fluorine and chlorine, have seven electrons in their outermost shells they tend to fill this shell by gaining an electron from other atoms, making them negatively-charged ions. Niels Bohr which after his PhD circa 1912 had joined Rutherford realized that. All of the negative charge was held in the electrons, orbiting the dense nucleus like planets around the sun. 
When an atom loses an electron to become a positively-charged ion, this is indicated by a plus sign after the element symbol for example, Na +. This model depicted an atomic model with nearly all its mass, and positive charge, in a central nucleus about 10,000 times smaller than the atom itself. Electrons have negative charge, protons have positive charge whereas neutrons have no charge. As a result of losing a negatively-charged electron, they become positively-charged ions. 1) An atom is made up of three particles: electrons, protons and neutrons. 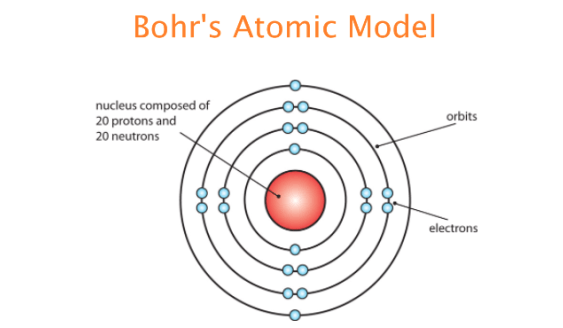
This means that they can achieve a stable configuration and a filled outer shell by donating or losing an electron. In comparison, the group 1 elements, including hydrogen (H), lithium (Li), and sodium (Na), all have one electron in their outermost shells. Their non-reactivity has resulted in their being named the inert gases (or noble gases). As shown in, the group 18 atoms helium (He), neon (Ne), and argon (Ar) all have filled outer electron shells, making it unnecessary for them to gain or lose electrons to attain stability they are highly stable as single atoms. Note that Bohr stated that electrons in the atom follow elliptical orbits (not. The periodic table is arranged in columns and rows based on the number of electrons and where these electrons are located, providing a tool to understand how electrons are distributed in the outer shell of an atom. In 1913, Danish physicist Niels Bohr applied Max Planck’s quantum theory to the nuclear atom of Ernest Rutherford, thus formulating the well-known planetary model of the atom, wherein electrons orbit a central nucleus in well-defined levels of energy ( Figure 1 ). Elements in other groups have partially-filled valence shells and gain or lose electrons to achieve a stable electron configuration.Īn atom may gain or lose electrons to achieve a full valence shell, the most stable electron configuration. A full valence shell is the most stable electron configuration. Group 18 elements (helium, neon, and argon are shown) have a full outer, or valence, shell. \):īohr diagrams indicate how many electrons fill each principal shell. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
